www.terumois.com/education and Step By Step Instructions
Overview Strictly for Educational/Informational Purposes in an Educational, Non-Clinical Setting:
The Terumo Angio-Seal VIP Vascular Closure Device (Ref. 610131) is a specialized medical instrument designed to quickly seal femoral artery punctures after catheterization procedures. It utilizes an 8 French (8F) system to achieve rapid hemostasis, significantly reducing the time patients need for bed rest and manual compression.
Device Components and Mechanism
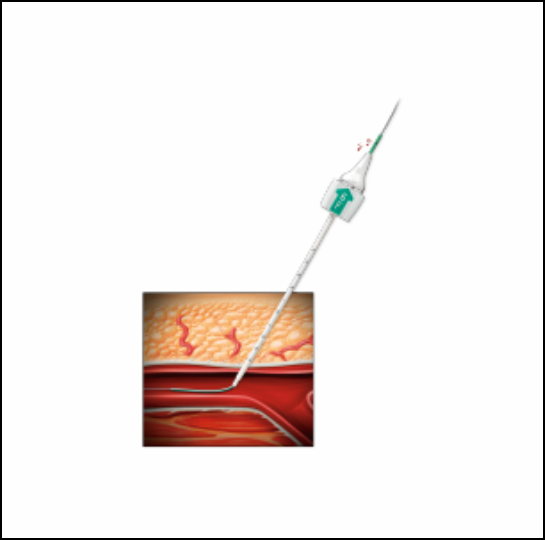
The Angio-Seal system creates a mechanical seal by "sandwiching" the arterial puncture (arteriotomy) between internal and external components. According to Terumo Interventional Systems, the system consists of:
Bioabsorbable Anchor: A polymer anchor placed against the inside of the vessel wall.
Collagen Sponge: Positioned on the exterior of the artery in the tissue tract to induce coagulation.
Self-Tightening Suture (STS): An absorbable suture that cinches the anchor and collagen together to form a secure, mechanical seal.
Absorption Timeline: All implanted components typically dissolve within 60 to 90 days.
Technical Specifications
Size: 8 French (8 Fr).
Guidewire Compatibility: 0.038 inches.
Anchor Dimensions: 10.3 x 1.96 x 1 mm.
Safety: The implanted components are MRI Safe and not made with natural rubber latex.
Clinical Usage and Indications
The device is indicated for use in patients who have undergone diagnostic angiography or interventional procedures using an 8F or smaller procedural sheath.
Terumo Interventional Systems
Primary Benefit: Allows for earlier ambulation (walking) and potential same-day hospital discharge compared to manual compression.
Contraindications: It should not be used in patients with known bovine collagen allergies or severe calcified arterial disease.
Q4.02242026gm